文章二维码:
如若转载,请注明e科网。
产品使用建议和意见可以联系小编进行优化处理:QQ:252188815、微信:13581664931
- 可见光催化;C-O活化;自由基化学;氟化学
如若转载,请注明e科网。
产品使用建议和意见可以联系小编进行优化处理:QQ:252188815、微信:13581664931
 清华成果发布 | 基于高精度地图和单目视觉传感器的车辆自定位技术孙悟空 2018-10-13
清华成果发布 | 基于高精度地图和单目视觉传感器的车辆自定位技术孙悟空 2018-10-13  【亿科创新智库】军民融合项目:新型无火药冷光焰火技术及其产业化巢博士 2018-04-04
【亿科创新智库】军民融合项目:新型无火药冷光焰火技术及其产业化巢博士 2018-04-04 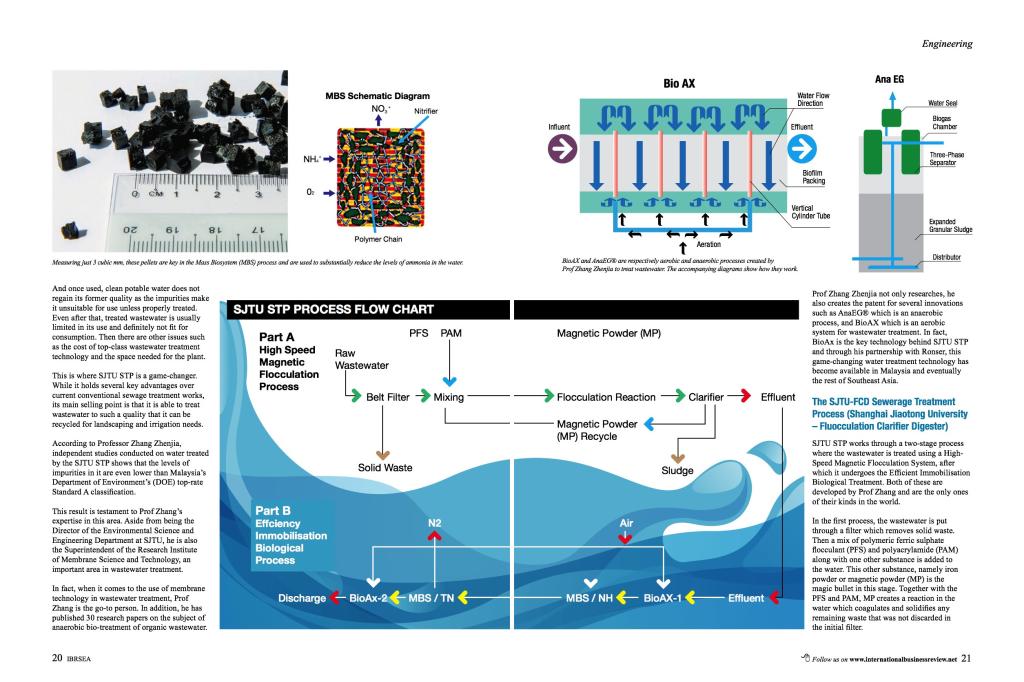 上海交大张振家团队“棕榈油废水零排放处理技术”金陵 2015-12-12
上海交大张振家团队“棕榈油废水零排放处理技术”金陵 2015-12-12  西交大在氢脆机理方面取得突破性进展,有望指导氢脆现象预防李明 2016-11-04
西交大在氢脆机理方面取得突破性进展,有望指导氢脆现象预防李明 2016-11-04 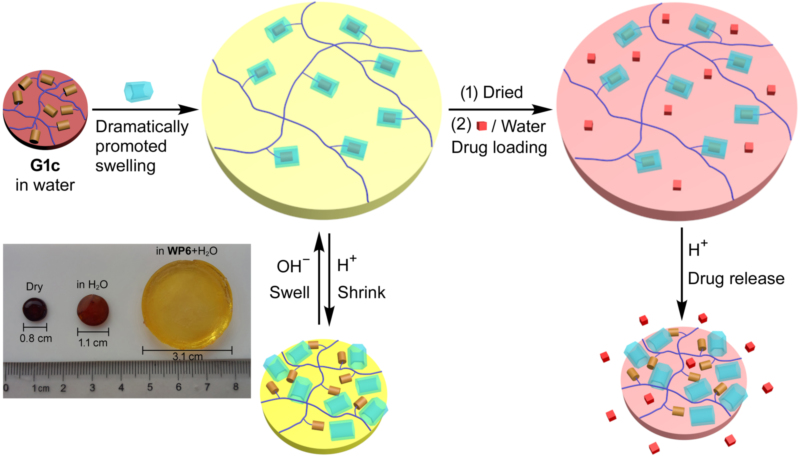 南京大学研究团队成功开发智能水凝胶,含水量提升12倍金陵 2016-05-17
南京大学研究团队成功开发智能水凝胶,含水量提升12倍金陵 2016-05-17  【亿科创新智库】生态农业项目:中国农大生态养殖技术及其产业化巢博士 2017-12-28
【亿科创新智库】生态农业项目:中国农大生态养殖技术及其产业化巢博士 2017-12-28 
发布成功!
确 定 关 闭